Apium M220
Apium provides a cost-effective solution to manufacture customized implants
The M220 is the world’s first 3D printer designed specifically for the manufacturing of medical products and implants made out of PEEK.
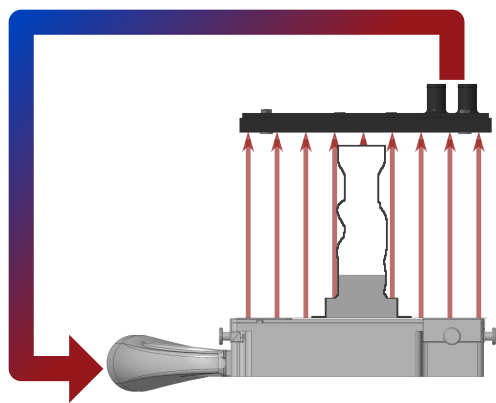
Sterile printing environment
The hot-air filter system analyses the component geometry and adapts the heating power so that an optimum energy input is always achieved. The air flow enclosing the component is filtered in a practically particle-free circuit.

New extrusion technology
Our extruder was developed regarding flexibility and durability. The four times higher feed force compared to the previous model enables a precise material flow. All components in contact with the printing material are made of medically compatible materials: PEEK, titanium, 316L steel and PTFE.

Biocompatible filaments
In cooperation with Evonik, materials and filaments were tested for their biocompatibility and the corresponding tests were successfully completed. This allows medical devices up to Class III to be manufactured from PEEK.
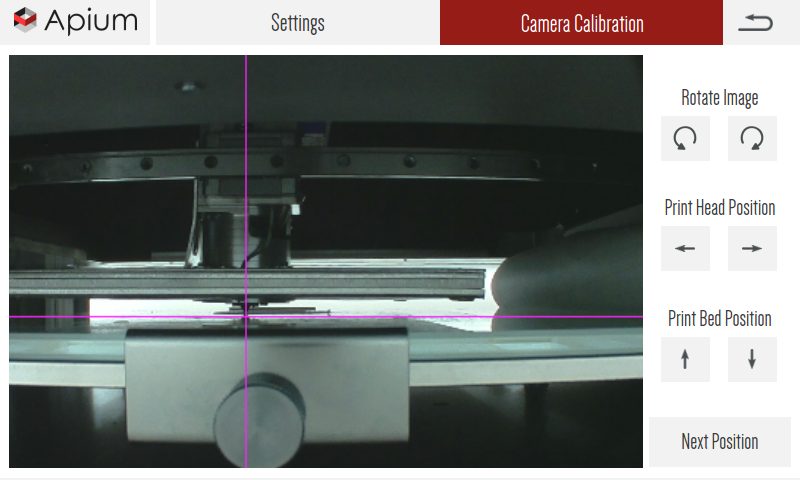
Monitored printing process
By integrating a camera system, temperature recording and servomotors, the printing process is continuously monitored. This enables the printer to intervene automatically when deviations occur.

Reproducibility
Integrated calibration routines guarantee consistent component quality. The software automatically informs about incoming maintenance operations and guides step-by-step through the procedures.

Documentation & Safty
All important parameters concerning the printing process are automatically provided in a PDF protocol. The integrated user administration prevents unwanted access. For your data security, we have decided against cloud based solutions.

Integrated software
The control software is specially adapted to our printer hardware and optimized for processing high-performance polymers. An intuitive user interface allows an easy handling of the printer. You can manage print jobs and check the status of the printer at any time.

Technical support
Our service department will train you in the handling of your printer. We will be happy to answer any questions you may have about the printer and other challenges of 3D printing.
Build Size
130 x 130 x 120 mm
Printer size
850 x 685 x 675 mm
Nozzle Diameter
0.2 – 0.8 mm
Layer Thicknesses
0.05 – 0.6 mm
Wall Thickness
> 250 µm
Printhead-Temperature
Up to 540°C
Hot Air Filter System
Up to 280°C
Print Bed Material
Stainless steel 316L
Noise Emission
< 70 dB(A)
Data Formats
STL, OBJ, G-Code
Life Saving Innovation
Based on the design freedom that comes along with our technology, complex geometries can be created on your desktop using radiological imaging data as input. The digital implant model, generated from the patient-specific data, can be manufactured from PEEK within a few hours, sterilized then deployed for patient treatment.
The opportunities which Apium’s Medical PEEK 3D Printing Technology offers, pertains most to the craniomaxillofacial, hand and spine areas where patient specific solutions are in most demand. By implementing the Apium solution, accruable savings on time, manufacturing cost and personnel can be realized relative to traditional manufacturing solutions. The Apium solution makes possible a faster and more flexible reaction scenario at times of emergency.
Software is a strength of Apium
Software is one of Apium’s core competencies. With the user interface on the printer touchpad, Apium provides the user with simple and efficient operation. Special service and maintenance functions facilitate daily use of the printer and allow detailed access to all settings. The web interface application makes it easy to monitor printing processes with the integrated camera from a remote workplace and manage the printer via the network.
Frequently Asked Questions
Implantable medical PEEK material
Apium has developed M220 Series 3D printers for researching, developing and producing PEEK medical devices. We will supply natural colored, high viscosity polyether ether ketone (PEEK) Filament. This filament is specially designed for long term implantable medical devices.
We jointly research and develop PEEK polymer filaments with Evonik.
Learn more about medical grade PEEK filament here.
Biocompatibility
Apium M220 Series 3D printers ensure that the printing process does not affect the biocompatibility and sterilization characteristics of 3D printed PEEK medical devices. The biocompatibility of the medical grade PEEK Filament and 3D printed medical devices has been tested following ISO 10993 recommendations.
A summary of biocompatibility test results is available upon request.
Process workflow of 3D-printed PEEK medical devices
Apium M220 Series 3D printers usage in medical devices production is similar to other production technologies in medical industry.
In order to have a better understanding of the complete procedure please watch the video above this section or kindly read our article here.
Service and Support
Apium offers the unique solution to achieve high quality results with Additive Manufacturing of PEEK medical devices. In order to gain knowledge and skills about PEEK 3D printing, prior to your M220 machine delivery, you can start your experiments with P220 Series machines. An advanced training customized for your application area will be provided by our Service Center experts.
Through your Additive Manufacturing experience, Apium experts will support you with each step to your successful implementation and operation of Apium systems.
Procurement of the Apium M220 series
Procurement of medical PEEK filament
Medical grade PEEK Filament can only be purchased directly from Apium. In order to receive a quote, please contact Apium experts.
Straight to your own Apium M Series
Success Story of Patient Care: Plastic Surgery of Birth Defects
There is a certain variability within the physical formation of the human body. So each one of us develops a bit different in their structural shape and appearance. As soon as such variability in shape from birth on is accompanied by a handicap in daily life, one...
Success Story of Patient Care: Treatment of Accident Injuries
In this case the patient had met with an accident when she was 5years old. After the accident she was not able to open her mouth to speak. Back then, the doctors mentioned that the situation will improve in some months. But it was never improved and 27 years later she...