Additive Manufacturing for Medical Applications
Produce patient-specific medical implants, which are affordable and accessible for everyone in the world.
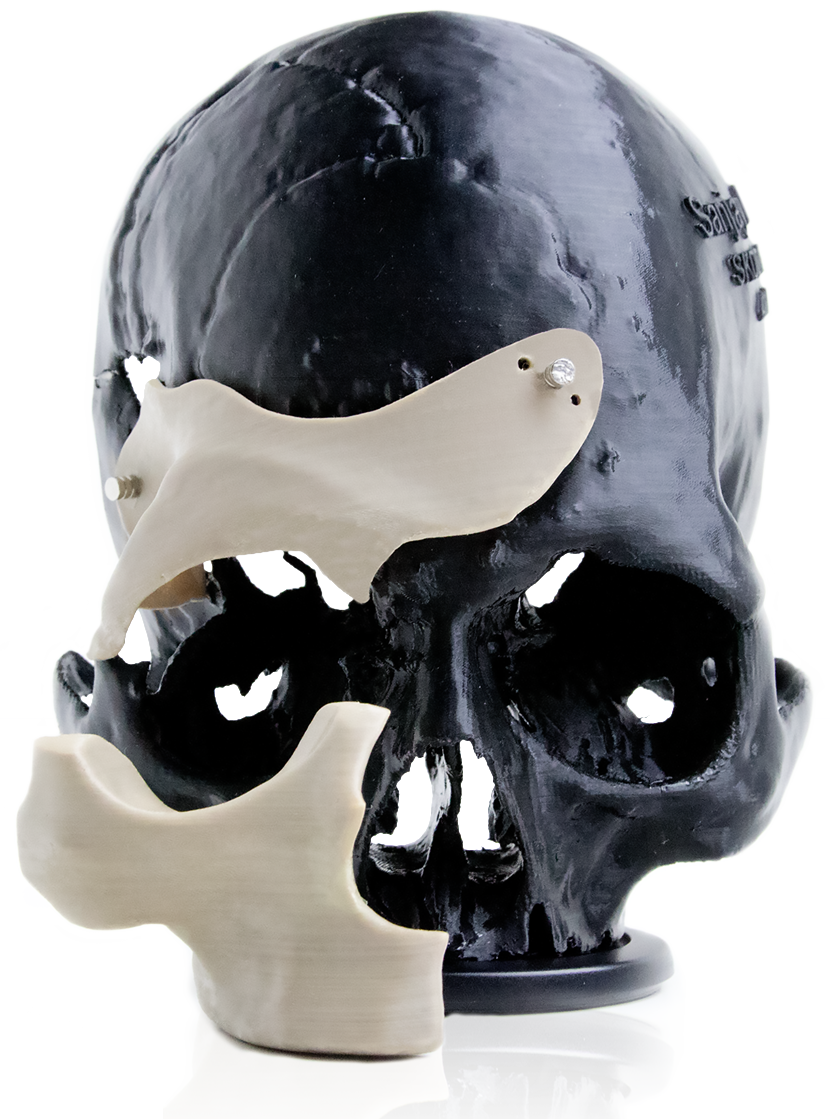
Part Design: Dr. Philipp Honigmann, Dr. Florian Thieringer
OSPIX: Sanjay Pathak, Global Health Care
Cost and Time Efficient Manufacture of Implants
Additive manufacturing is a cost and time saving process for single parts and small series production. By using the material extrusion process technology, the Apium M220 makes it now possible to manufacture implants and prosthetics. The thermoplastic PEEK (polyether-ether-ketone) has established its position as a suitable material for long-term implants and it is ideal for the material extrusion process in order to manufacture mechanically resilient parts.
Conventionally, PEEK implants are milled which produces material waste by nature. In the following case study, it is shown that 3D printing PEEK for medical application is not only material efficient but also reduces time and cost.

Design, Slice and Print with M220
Our 3D printing technology can be easily integrated into an existing medical workflow. Data used to reconstruct defects are converted into a machine code that results in component generation by the M220.
Manufacturing Advantages with Material Extrusion Technology
Using the example of a patient-specific skull implant, the time and cost savings compared to a conventionally manufactured implant are demonstrated.
First of all, the skull implant was loaded with 2.99 kN on a small area before the component cracked. A human skull withstands a force of 2.88 kN in a comparable test [1].
[1] C. Van Lierde, B. Depreitere, J. Vander Sloten, R. Van Audekercke, G. Van Der Perre und J. Goffin, „Skull biomechanics: The energy absorbability of the human skull frontal bone during fracture under quasistatic loading,“ Journal of Applied Biomaterials & Biomechanics, 2003.



Apium M220 3D Printing Technology vs Milling Technology
Comparing the manufacturing cost and time for the same skull implant produced by milling and 3D printing, the results of this case study show that 3D printing technology provides:
- 89% material reduction
- 73% cost reduction
Additionally, parts produced with an Apium M220 have the following benefits:
- ISO 13485 includable
- Medical devices grade III producible
- Detailed print protocols for quality control
*The milling information has been provided by SRM AG.
Publications Related on Medical Applications with 3D Printing
Patient specific Implants from a 3D printer – An Innovative Manufacturing Process for Custom PEEK Implants in Cranio-Maxillofacial Surgery
(Florian Thieringer, Neha Sharma, Azagen Mootien, Ralf Schumacher, Philipp Honigmann – 978-3-319-66865-9)
Cranial polyetheretherketone implants by extrusion-based additive manufacturing: state of the art and prospects
(Matthias Katschnig, Clemens Holzer – 10.15406/mseij.2018.02.00036)
Structure-Property Relationships for 3D printed PEEK Intervertebral Lumbar Cages Produced using Fused Filament Fabrication
(Cemile Basgul, Tony Yu, Daniel W. MacDonald, Ryan Siskey, Michele Marcolongo, Steven M. Kurtz – 10.1557/jmr.2018.178)
Patient-Specific Surgical Implants Made of 3d Printed PEEK: Material, Technology, and Scope of Surgical Application
(Philipp Honigmann, Neha Sharma, Brando Okolo, Uwe Popp, Bilal Msallem, Florian Thieringer – 10.1155/2018/4520636)
Performance of biocompatible PEEK processed by fused deposition additive manufacturing
(M.F. Arif, S. Kumar, K.M. Varadarajan, W.J. Cantwell – 10.1016/j.matdes.2018.03.015)
No Results Found
The page you requested could not be found. Try refining your search, or use the navigation above to locate the post.
Apium M220 Technology
Explore our innovative technology for medical applications.
More About
Honour us with researching on medical peek printing technologies.
